|
Red fluorescent protein FusionRed
- Superior performance in fusions
- Low cytotoxicity
- Fast maturation, high pH-stability and photostability
- Proven suitability to generate stably transfected cell lines
- Recommended for protein labeling and long-term experiments
 |
FusionRed is a red fluorescent protein characterized by improved performance in fusions and low toxicity [Shemiakina et al., 2012]. FusionRed lacks the residual tendency of other monomeric RFPs to dimerize at high concentration and behaves as a pure monomer at concentrations up to 10 mg/ml in HPLC analysis. Such "supermonomeric" properties ensure superior efficiency of FusionRed in protein labeling applications, especially in the cells with high expression level.
Similarly to parental mKate2, FusionRed demonstrates fast maturation rate, high pH-stability and photostability, and significantly lower cytotoxicity than widely used mCherry [Shaner et al., 2004] and mRuby [Kredel et al., 2009].
FusionRed is mainly intended for protein labeling and long-term experiments including generation of transgenic animals.
|
Main properties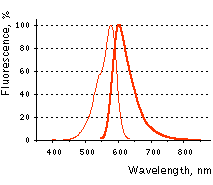
FusionRed normalized excitation (thin line) and emission (thick line) spectra.
Spectra viewer tool
Download FusionRed spectra (xls)
| | CHARACTERISTIC | |
|---|
* Brightness is a product of extinction coefficient and quantum yield, divided by 1000.
** Purified recombinant protein behaves as a pure monomer at concentrations of 10 mg/ml as verified by high-performance liquid chromatography (HPLC)
*** Time to bleach 50% of fluorescent signal brightness.
| | Molecular weight, kDa | 26 | | Polypeptide length, aa | 232 | | Fluorescence color | red | | Excitation maximum, nm | 580 | | Emission maximum, nm | 608 | | Quantum yield | 0.19 | | Extinction coefficient, M-1cm-1 | 94 500 | | Brightness* | 18.0 | | Brightness, % of EGFP | 53 | | pKa | 4.6 | | Structure | supermonomer** | | Aggregation | no | | Maturation half-time, min | 130 | | Photostability, widefield*** | 150 | | Photostability, confocal*** | 176 | | Cell toxicity | not observed | | Possible limitations | superior performance in fusions, low cytotoxicity |
|
|---|
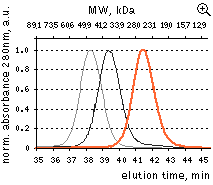 | HPLC analysis of FusionRed in comparison with selected fluorescent proteins loaded in a high concentration.
FusionRed – orange line, mKate2 – black line, mNeptune – gray line. Proteins were loaded at a concentration of 10 mg/ml. While HPLC demonstrates reversible dimerization of mKate2 and reveals a dimeric character for the mKate derivative, far-red fluorescent protein mNeptune [Lin et al., 2009], FusionRed behaves as a pure monomer. Data from Shemiakina et al., 2012
|
|---|
Recommended filter sets and antibodies
FusionRed can be recognized using Anti-tRFP antibody (Cat.# AB233) available from Evrogen.
FusionRed can be detected using TRITC filter set or similar. Recommended Omega Optical filter sets are QMAX-Red and XF102-2.
Performance and use
FusionRed can be easily expressed and detected in a wide range of organisms. Mammalian cells transiently transfected
with FusionRed expression vectors produce bright fluorescence in 10-12 hours after transfection. FusionRed
performance in fusions has been demonstrated in a number of models.

|
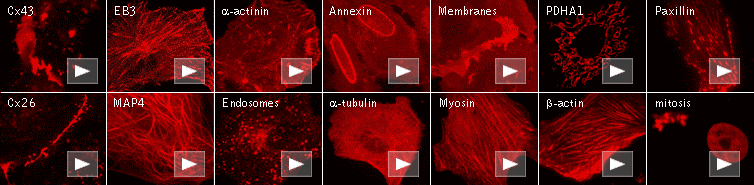
FusionRed use for protein labeling in mammalian cells.
Scale bars represent 10 μm. Data from Shemiakina et al., 2012
|
Labeling efficiency testing
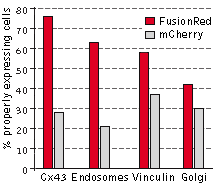 |
The performance of FusionRed and mCherry was directly compared in HeLa CCL2 cells for the following
targets: connexin-43, endosomes, vinculin, and the Golgi complex. In this experiment, the cells were
transfected with each of the four target fusions, fixed, and then compared for localization efficiency,
which was determined by calculating the percentage of properly expressing cells versus the total number
of transfected cells. FusionRed demonstrated clear advantage in all four fusions. Data from Shemiakina
et al., 2012
|
Cytotoxicity testing in HeLa cells
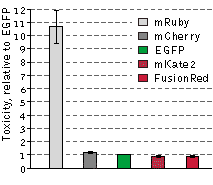 |
The cytotoxicity of FusionRed relative to selected red fluorescent proteins and EGFP was evaluated in
the following experiment: HeLa cells were transfected with appropriate vectors encoding EGFP or one of
the following red fluorescent proteins: mRuby, FusionRed, mKate2 or mCherry. Next, the EGFP-expressing
cells were mixed with those expressing one of the RFPs, resulting in 4 separate cell mixtures: EGFP and
mRuby, EGFP and FusionRed, EGFP and mKate2, and EGFP and mCherry. 48 hours after transfection, the
green-to-red cell ratios were calculated utilizing flow cytometry and each of the cell mixtures were
then plated into 3 plates. After additional 92 hour incubation, the green-to-red cell ratios were
recalculated. Because only living cells were counted for this experiment, the difference between the
ratios before and after the incubation can be assumed to accurately reflect RFP toxicity versus EGFP.
mRuby exhibited a more than 10-fold higher cytotoxicity level compared to EGFP, while the remaining RFPs
were almost as cytotoxic as EGFP in this experiment. Data from Shemiakina et al., 2012
|
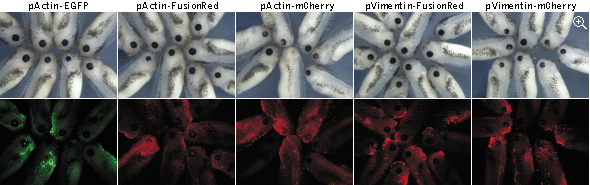
Cytotoxicity testing in Xenopus laevis tadpoles
The eye formation is a complex multistage process
that depends on many mechanisms, any disturbance of which may result in an abnormal phenotype. Here eye development
was used as a sensitive readout of possible abnormal course of embryogenesis caused by expression of EGFP,
FusionRed, and mCherry fusions. In these experiments, actin and vimentin fusions of EGFP and FusionRed were only
slightly more toxic than the uninjected controls, while expression of mCherry fusions led to notable decrease of the
average eye size. Moreover, in many cases (80% of tadpoles with the reduced eye size), the reduced eye phenotype was
accompanied by varying degree of an unclosed optic fissure, indicating an abnormal eye development.
|
Stage 41 Xenopus laevis tadpoles expressing fluorescent protein fusions. Top panels: white light;
bottom panels: fluorescence. Data from Shemiakina et al., 2012
|
 |
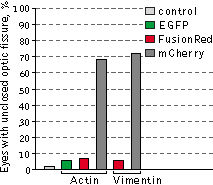 |
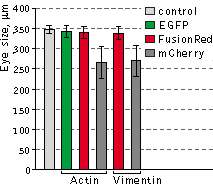
Left graph: Mean values of eye size of Xenopus laevis embryos injected with plasmids expressing
different fluorescent protein fusions.
Right graph: Percentage of reduced eyes with unclosed optic fissure.
Data from Shemiakina et al., 2012
|
Long-term expression
The excellent suitability of FusionRed for long-term experiments was proved both in
experiments with stably transfected cell lines and in transgenic animals. In addition to its low cytotoxicity,
FusionRed does not show abnormal lysosomal localization typical for many fluorescent proteins in long-term
expression.
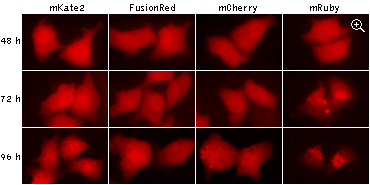
|
Being expressed in HeLa cells, both mKate2 and FusionRed remain evenly localized in cytoplasm after 96
hours of cultivation, while mCherry and mRuby show abnormal lysosomal localization. Data from Shemiakina
et al., 2012
|
Available variants and fusions
| Variant | Description | Related vector | Cat.# | Click for image |
|---|
 |
|
Humanized FusionRed
|
FusionRed codon usage is optimized for high expression in mammalian cells [Haas et al., 1996], but it can be successfully expressed in many other heterological systems.
|
pFusionRed-N
|
FP412
|
|
FusionRed-Cx43 fusion
|
Rat connexin 43 is fused to the FusionRed N-terminus. When expressed in mammalian cells, this fusion provides red fluorescent labeling of connexin 43 in living cells.
|
pFusionRed-Cx43
|
FP417
|
|
|
FusionRed-f-mem fusion
|
20 amino acid farnesylation signal from c-Ha-Ras is fused to the FusionRed C-terminus. When expressed in mammalian cells, this fusion provides red fluorescent labeling of plasma membrane.
|
pFusionRed-f-mem
|
FP418
|
|
|
FusionRed-H2B fusion
|
Human histone H2B is fused to the FusionRed N-terminus. When expressed in mammalian cells, this fusion provides red fluorescent labeling of histone H2B in living cells.
|
pFusionRed-H2B
|
FP421
|
|
|
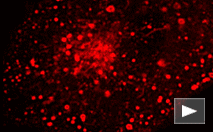
FusionRed-endo fusion
|
Human RhoB GTPase is fused to the FusionRed C-terminus. When expressed in mammalian cells, this fusion provides red fluorescent labeling of vesicles of the endocytic pathway.
|
pFusionRed-endo
|
FP427
|
|
|
FusionRed-PDHA1 fusion
|
Human pyruvate dehydrogenase alpha 1 (PDHA1) is fused to the FusionRed N-terminus. When expressed in mammalian cells, this fusion provides red fluorescent labeling of PDHA1 in living cells.
|
pFusionRed-PDHA1
|
FP430
|

|
|
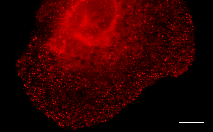
FusionRed-Rab5a fusion
|
Human Ras-related protein Rab-5A is fused to the FusionRed C-terminus. When expressed in mammalian cells, this fusion provides red fluorescent labeling of Rab-5A in living cells.
|
pFusionRed-Rab5a
|
FP431
|
|
|
FusionRed-talin fusion
|
Mouse talin-1 protein is fused to the FusionRed N-terminus. When expressed in mammalian cells, this fusion provides red fluorescent labeling of talin in living cells.
|
pFusionRed-talin
|
FP432
|
References:
-
Fliegel L, Burns K, MacLennan DH, Reithmeier RA, Michalak M.
Molecular cloning of the high affinity calcium-binding protein (calreticulin) of skeletal muscle sarcoplasmic reticulum.
J Biol Chem. 1989; 264 (36):21522-8. / pmid: 2600080
-
Haas J, Park EC, Seed B.
Codon usage limitation in the expression of HIV-1 envelope glycoprotein.
Curr Biol. 1996; 6 (3):315-24. / pmid: 8805248
-
Kredel S, Oswald F, Nienhaus K, Deuschle K, Röcker C, Wolff M, Heilker R, Nienhaus GU, Wiedenmann J.
mRuby, a bright monomeric red fluorescent protein for labeling of subcellular structures.
PLoS One. 2009; 4 (2):e4391. doi: 10.1371/journal.pone.0004391 / pmid: 19194514
-
Lin MZ, McKeown MR, Ng HL, Aguilera TA, Shaner NC, Campbell RE, Adams SR, Gross LA, Ma W, Alber T, Tsien RY.
Autofluorescent proteins with excitation in the optical window for intravital imaging in mammals.
Chem Biol. 2009; 16 (11):1169-79. doi: 10.1016/j.chembiol.2009.10.009 / pmid: 19942140
-
Munro S, Pelham HR.
A C-terminal signal prevents secretion of luminal ER proteins.
Cell. 1987; 48 (5):899-907. / pmid: 3545499
-
Shaner NC, Campbell RE, Steinbach PA, Giepmans BN, Palmer AE, Tsien RY.
Improved monomeric red, orange and yellow fluorescent proteins derived from Discosoma sp. red fluorescent protein.
Nat Biotechnol. 2004; 22 (12):1567-72. / pmid: 15558047
-
Shemiakina II, Ermakova GV, Cranfill PJ, Baird MA, Evans RA, Souslova EA, Staroverov DB, Gorokhovatsky AY, Putintseva EV, Gorodnicheva TV, Chepurnykh TV, Strukova L, Lukyanov S, Zaraisky AG, Davidson MW, Chudakov DM, Shcherbo D.
A monomeric red fluorescent protein with low cytotoxicity.
Nat Commun. 2012; 3 :1204. doi: 10.1038/ncomms2208 / pmid: 23149748
|