
|
||||||||||
|
||||||||||

Katushka2S
SUPPORTRESOURCES |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Katushka2S is the next generation of far-red fluorescent protein TurboFP635 (Katushka) [Luker et al., 2015; Shcherbo et al., 2009; Gurskaya et al., 2011]. Katushka2S exhibits highest brightness, fastest maturation and superior signal-to-noise ratio compared to TurboFP635 and many other available far-red fluorescent proteins: E2-Crimson, mNeptune, mNeptune2.5, mCardinal, TurboFP650 and NirFP [Luker et al., 2015], which makes it the protein of choice for visualization within living tissues. Katushka2S can also be used together with iRFP720 [Luker et al., 2015] for dual color whole body imaging. |
Main properties
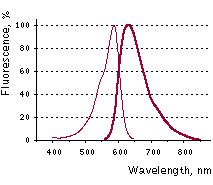
Katushka2S normalized excitation (thin line) and emission (thick line) spectra. |
| ||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Recommended filter sets and antibodies
Katushka2S can be recognized using Anti-tRFP antibody (Cat.# AB233) available from Evrogen.
Recommended Omega Optical filter sets are QMAX-Red and XF102-2. Katushka2S can also be detected using Texas Red filter sets or similar.
For IVIS Lumina II imaging system the highest fluorescence signal for Katushka2S is observed with the following settings:
In cell culture: "Cy5.5" (695-770 nm) channel using 570/35nm excitation
In whole body imaging: "Cy5.5" (695-770 nm) channel using 605/35nm excitation
Performance and use
Mammalian cells transiently transfected with Katushka2S expression vectors produce bright fluorescence in 12 hrs after transfection. No cytotoxic effects or visible protein aggregation are observed.
Superior performance of Katushka2S in whole-body imaging was demonstrated using mouse xenograft model. HEK293FT cells transiently transfected with plasmids encoding different far-red fluorescent proteins were implanted into mice intramuscularly or subcutaneously. The cells were co-transfected with firefly luciferase to normalize the transfection efficiency and total numbers of injected cells. Katushka2S produced higher fluorescence signal and better signal-to-noise ratio than other far-red fluorescence proteins analyzed at various excitation and emission channels.
 |  | |
|---|---|---|
|
| ||
 |  | |
|---|---|---|
|
| ||
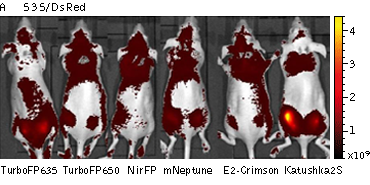
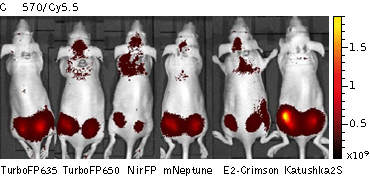
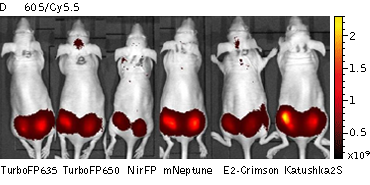
Whole-mouse imaging with IVIS Lumina II system (PerkinElmer).Representative fluorescence images of nude mice injected into the gluteal muscle with HEK293FT cells transiently expressing mNeptune, E2-Crimson, Katushka2S, TurboFP635, TurboFP650 and NirFP captured with indicated excitation (35-nm bandwidth centered at the indicated wavelength) and emission (DsRed 575-650nm and Cy5.5 695-770nm) filter combination. Pseudocolor scale bar: radiant efficiency (photons/s)/(µW/cm2). Note that scale bar differ for images with different excitation and emission filters. Images from [Luker et al., 2015] |
 |  |  | |
|---|---|---|---|
|
| |||
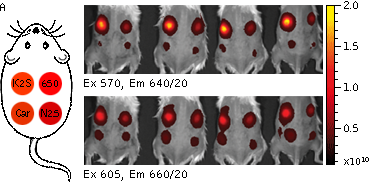
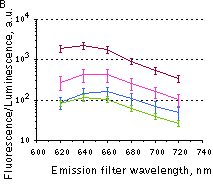
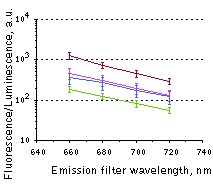
Whole-mouse imaging with IVIS Spectrum system (PerkinElmer).(A) Representative fluorescence images of mice injected in backs with HEK293FT cells transiently expressing Katushka2S (K2S), TurboFP650 (650), mCardinal (Car) and mNeptune2.5(N2.5). captured with indicated excitation (35-nm bandwidth centered at the indicated wavelength) and emission (640/20 and 660/20) filter combination. Pseudocolor scale bar: radiant efficiency (photons/s/cm2/sr)/(µW/cm2). (B) Graphs display mean values ±SEM for fluorescence radiant efficiency normalized to luciferase photon flux for each implant (n=4 per condition) for 570-nm (left) and 605-nm (right) excitation and listed emission filters. Fluorescent proteins are depicted by the following colors: Katushka2S (brown), TurboFP650 (pink), mCardinal (blue) and mNeptune2.5 (green). Images from [Luker et al., 2015] |
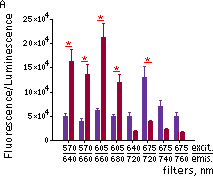
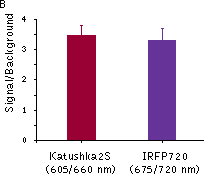
Katushka2S was also compared with one of the best phytochrome photoreceptors iRFP720. Katushka2S produced brighter fluorescence intensity at its optimum conditions as compared with the best output from iRFP720. At the same time, both proteins showed comparable signal-to-noise ratios at the optimum wavelengths for excitation and emission since autofluorescence from mouse tissue was lower in the channel optimal for iRFP720.
 |  | |
|---|---|---|
|
| ||
Fluorescence signals acquired with IVIS Spectrum system (PerkinElmer).Fluorescence signals normalized to corresponding firefly luciferase luminescence in mice injected in backs with HEK293FT cells transiently expressing Katushka2S or iRFP70. (A) Pairwise comparisons of the signals from Katushka2S (brown) and iRFP720 (violet) at different excitation/emission wavelengths. * p<0.0005 (B) Signal-to-noise ratios at optimal excitation and emission wavelengths for each protein. |
Note: Katushka2S demonstrates lower photostability than its parental variant TurboFP635. It may limit Katushka2S utility in cell culture experiments, but not in whole body imaging, where scattering and absorption of light naturally limit excitation power [Leblond et al., 2010].
| Variant | Description | Related vector | Cat.# | |
|---|---|---|---|---|
 | ||||
| Humanized Katushka2S | Katushka2S codon usage is optimized for high expression in mammalian cells [Haas et al., 1996], but it can be successfully expressed in many other heterological systems. |
|
FP761 | |
|
|
FP763 | |||
References:
- Gurskaya NG, Staroverov DB, Fradkov AF, Lukyanov KA. Coding region of far-red fluorescent protein katushka contains a strong donor splice site. Bioorg Khim. 2011; 37 (3):425-8. (in Russian) / pmid: 21899059
- Haas J, Park EC, Seed B. Codon usage limitation in the expression of HIV-1 envelope glycoprotein. Curr Biol. 1996; 6 (3):315-24. / pmid: 8805248
- Leblond F, Davis SC, Valdes PA, Pogue BW. Pre-clinical whole-body fluorescence imaging: Review of instruments, methods and applications. J Photochem Photobiol B. 2010; 98 (1):77-94. doi: 10.1016/j.jphotobiol.2009.11.007 / pmid: 20031443
- Luker KE, Pata P, Shemiakina II, Pereverzeva A, Stacer AC, Shcherbo DS, Pletnev VZ, Skolnaja M, Lukyanov KA, Luker GD, Pata I, Chudakov DM. Comparative study reveals better far-red fluorescent protein for whole body imaging. Sci Rep. 2015; 5 :10332. doi: 10.1038/srep10332 / pmid: 26035795
- Shcherbo D, Murphy CS, Ermakova GV, Solovieva EA, Chepurnykh TV, Shcheglov AS, Verkhusha VV, Pletnev VZ, Hazelwood KL, Roche PM, Lukyanov S, Zaraisky AG, Davidson MW, Chudakov DM. Far-red fluorescent tags for protein imaging in living tissues. Biochem J. 2009; 418 (3):567-74. doi: 10.1042/BJ20081949 / pmid: 19143658
|
Copyright 2002-2023 Evrogen. All rights reserved. Evrogen JSC, 16/10 Miklukho-Maklaya str., Moscow, Russia, Tel +7(495)988-4084, Fax +7(495)988-4085, e-mail:evrogen@evrogen.com |



