|
Photoactivatable red fluorescent protein PA-TagRFP
- Monomer, successful performance in fusions
- Non-fluorescent before photoactivation
- Irreversible photoactivation to a red fluorescent form by UV-violet light irradiation
- High brightness and photostability
- Recommended for super-resolution imaging
 |
PA-TagRFP is a photoactivatable mutant of the bright monomeric red fluorescent protein TagRFP [Subach et al., 2010]. PA-TagRFP is capable of irreversible photoconversion from non-fluorescent to red fluorescent form (with excitation/emission maxima at 562 nm and 595 nm, respectively) in response to UV-violet light irradiation.
High brightness, photostability and monomeric nature of PA-TagRFP make it an excellent protein tag for both conventional microscopy and super-resolution PALM imaging techniques [Subach et al., 2010].
|
Main properties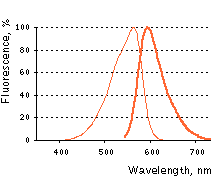
Normalized excitation (thin line) and emission (thick line) spectra for activated PA-TagRFP.
Download PA-TagRFP spectra (xls)
| | CHARACTERISTIC | before / after photoactivation |
|---|
* Brightness is a product of extinction coefficient and quantum yield, divided by 1000.
| | Fluorescence color | NO / red | | Excitation maximum, nm | - / 562 | | Emission maximum, nm | - / 595 | | Quantum yield | nd / 0.38 | | Extinction coefficient, M-1cm-1 | nd / 66 000 | | Brightness* | 0 / 25.1 | | pKa | nd / 5.3 | | Activating light | UV-violet (e.g. 390-420 nm) | | Calculated contrast, fold | ~540 | | Structure | monomer | | Cell toxicity | not observed | | Aggregation | no | | Maturation rate at 37°C | fast | | Molecular weight, kDa | 27 | | Polypeptide length, aa | 237 |
|
|---|
Recommended antibodies, filter sets and laser lines
PA-TagRFP can be recognized using Anti-tRFP antibody (Cat.# AB233) available from Evrogen.
PA-TagRFP is non-fluorescent before light activation. Upon UV-violet irradiation the protein irreversibly converts to its red fluorescent form.
PA-TagRFP can be activated during both widefield imaging (e.g. the Arc-lamp irradiation, 100xoil objective, 390-420 nm, 10-50 mW/cm2) and confocal laser scanning imaging (e.g. 405 nm laser line, estimated < 2.5 W/cm2 at the sample). Maximal efficiency of photoactivation for PA-TagRFP is observed at 390-420 nm. The photoactivation efficiency drops dramatically with the wavelength increasing above 420 nm.
The source of irradiation, irradiation time and intensity of activating UV-violet light must be individually adjusted for particular instrumentation and intended application.
TRITC filter set or similar can be used for visualization of activated PA-TagRFP. Omega Optical filter sets QMAX-Red and XF174 are recommended.
Performance and use
PA-TagRFP can be easily expressed and detected in a wide range of organisms. Mammalian cells transiently transfected with PA-TagRFP expression vectors produce bright fluorescence upon UV-activation of PA-TagRFP in 10-12 hrs after transfection. No cytotoxic effects or visible protein aggregation are observed.
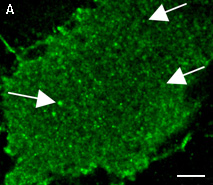
 | PA-TagRFP use for cell labeling.
Live HeLa cells transiently transfected with the PA-TagRFP-C expression vector were imaged during the photoactivation.
|
|---|
PA-TagRFP performance in protein fusions has been demonstrated in β-actin, α-tubulin, histone H2B and other models.
PA-TagRFP use in PALM imaging techniques
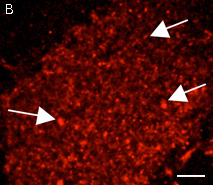
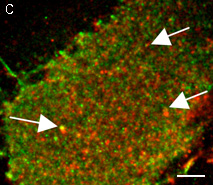
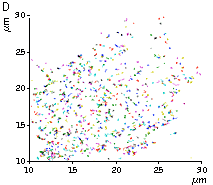
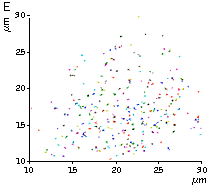
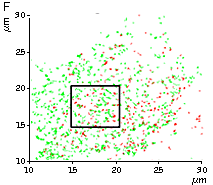
High brightness, photostability and absence of initial fluorescence signal from PA-TagRFP make it a protein tag of choice for super resolution two-color PALM/single-particle tracking PALM imaging techniques. The excellent performance of PA-TagRFP in two-color single-particle tracking PALM experiments was demonstrated for several PA-TagRFP-tagged and PAGFP-tagged fusions in live COS-7 cells [Subach et al., 2010].
An example for the tracking of PA-TagRFP-tagged epidermal growth factor receptor (EGFR-PATagRFP) and PAGFP-tagged vesicular stomatitus virus G protein tsO45 (VSVG-PAGFP) in live COS-7 cells by two-color single-particle tracking PALM is shown below.
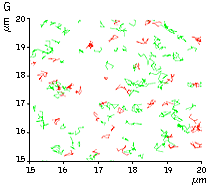 |
(G) A zoomed view of the region indicated by the square in (F).
|
|---|
Available variants and fusions
| Variant | Description | Related vector | Cat.# |
|---|
 |
|
Humanized PA-TagRFP
|
PA-TagRFP codon usage is optimized for high expression in mammalian cells [Haas et al., 1996], but it can be successfully expressed in many other heterological systems.
|
pPA-TagRFP-C
|
FP811
|
|
pPA-TagRFP-N
|
FP812
|
|
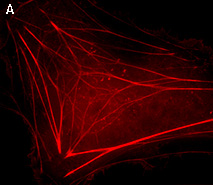
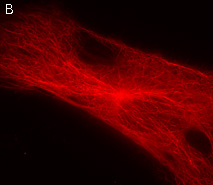
PA-TagRFP-tubulin fusion
|
Human α-tubulin is fused to the PA-TagRFP C-terminus. When expressed in mammalian cells, this fusion provides red fluorescent labeling of α-tubulin in living cells.
|
pPA-TagRFP-tubulin
|
FP814
|
|
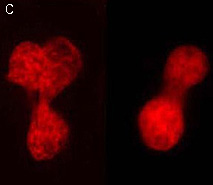
PA-TagRFP-H2B fusion
|
Human histone H2B is fused to the PA-TagRFP N-terminus. When expressed in mammalian cells, this fusion provides red fluorescent labeling of histone H2B in living cells.
|
pPA-TagRFP-H2B
|
FP815
|
References:
-
Haas J, Park EC, Seed B.
Codon usage limitation in the expression of HIV-1 envelope glycoprotein.
Curr Biol. 1996; 6 (3):315-24. / pmid: 8805248
-
Subach FV, Patterson GH, Renz M, Lippincott-Schwartz J, Verkhusha VV.
Bright monomeric photoactivatable red fluorescent protein for two-color super-resolution sptPALM of live cells.
J Am Chem Soc. 2010; 132 (18):6481-91. doi: 10.1021/ja100906g / pmid: 20394363
|