
|
Caspase-3 apoptosis sensor Casper3-GR
- Early detection of Caspase-3 activity onset
- High sensitivity
- Direct expression in cells
- No exogenous chemical compounds required
- Proven suitability for FLIM-based screenings
- Recommended for early detection of apoptosis
Performance and use
The excellent performance of Casper3-GR sensor has been demonstrated in vivo on staurosporine-induced apoptosis of HeLa cells [Shcherbo et al., 2009]. Living cells were monitored at 37°C with Leica SP2 confocal microscope (excitation using 488 nm laser line, emission collected at 500-530 nm and 560-650 nm). The fluorescence was evenly distributed in the cytosol and nucleus with no aggregation or non-specific localization observed. Importantly, both green and red signals were reliably stable under various irradiation conditions for hours. No reversible or irreversible fluorescence bleaching or photoconversion was observed.
Approximately 30-40 min after 2 μM staurosporine infusion, cells demonstrated rapid (within 10 min) and pronounced changes in green-to-red fluorescence signal ratio, indicating activation of caspase-3. Later these cells demonstrated characteristic membrane blebbing. The average contrast in living cells (calculated as donor/acceptor emission ratio change for 5 cells, time point aligned to the median of ratio changes, individual for each cell) reached 3.8-fold.
Measurement of Casper3-GR apoptosis induced FRET changes by FLIM revealed the dramatic increase of TagGFP fluorescence lifetime from 1.5 ns to 2.5 ns. The FRET efficiency of the uncleaved Casper3-GR (38% based on the phase lifetime) is among the highest measured by FLIM. Since the FRET efficiency of the cleaved substrate is zero, the dynamic range of the sensor is rather high, indicating that Casper3-GR can be successfully used for the high content FLIM based screenings on living cells.
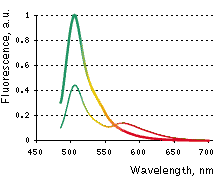 | Emission spectra of Casper3-GR before (thin line) and after digestion by Caspase-3 (thick line).
|
|---|
 |  |  | |
|---|
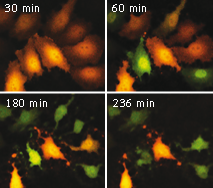
Casper3-GR upon staurosporine-induced apoptosis in HeLa cells.
Two channel fluorescence imaging of Casper3-GR upon staurosporine-induced apoptosis in HeLa cells. Time, in minutes, is shown after staurosporine infusion.
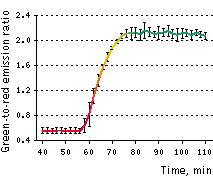
On the left graph, green-to-red emission ratio change of Casper3-GR upon staurosporine-induced apoptosis. Approximately 40-50 min after staurosporine infusion, cells demonstrated pronounced changes fluorescence signal ratio. Emission ratio shown for 5 cells, time point aligned to the median of ratio changes, individual for each cell. Excitation at 488 nm, emission was detected at 500-530 nm and 560-600 nm.
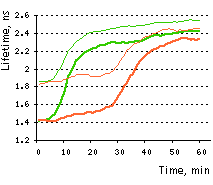
On the right graph, TagGFP fluorescence phase lifetime (thick lines) and average modulation lifetime (thin lines) changes for Casper3-GR during staurosporine-induced apoptosis. Excitation was at 488 nm and donor fluorescence emission was passed through a 500-530 nm bandpass filter.
|
References:
-
Shcherbo D, Souslova EA, Goedhart J, Chepurnykh TV, Gaintzeva A, Shemiakina II, Gadella TW, Lukyanov S, Chudakov DM.
Practical and reliable FRET/FLIM pair of fluorescent proteins.
BMC Biotechnol. 2009; 9 :24. doi: 10.1186/1472-6750-9-24 / pmid: 19321010
|









