
|
||||||||||
|
||||||||||

|
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
The vector sequence has been compiled using the information from sequence databases, published literature, and other sources, together with partial sequences obtained by Evrogen. This vector has not been completely sequenced. |
|||||||||||||||||||||||||||||||||||||||||||||||||
 Download
|
| ||||||||||||||||||||||||||||||||||||||||||||||||
| Nhe I | Bgl II* | Sac I | EcoR I* | Sal I | Sac II | Sma I/Xma I | Age I | TurboGFP-dest1 | ||||||||||||||||||||||||
| Afe I | Xho I* | Hind III | Pst I* | Kpn I | Apa I* | BamH I* | Nco I* | |||||||||||||||||||||||||
| ... | G.CT | A.GC | G.CT | A.CCG.GAC.TC | A.GAT. | CT | C. | GAG | .CTC | . | AAG.CTT | .C | GA.ATT. | C | TG.CA | G | .TCG.AC | G.GTA. | CC | G.C | GG | .G | CC.C | G | G.G | AT.CC | A.CCG.GT | C.GCC.A | CC. | ATG.G | ... | |
* – not unique site.
Vector description
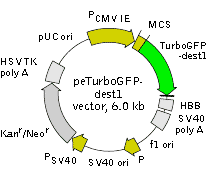
peTurboGFP-dest1 is a mammalian expression vector encoding destabilized variant of the green fluorescent protein TurboGFP (see reporter description). To generate TurboGFP-dest1 variant, residues 422-461 of mouse ornithine decarboxylase (MODC) were fused to the TurboGFP C-terminus. This MODC region contains a PEST amino acid sequence that targets the protein for degradation and provides for rapid protein turnover [Li et al., 1998]. TurboGFP-dest1 retains fluorescent properties of the native protein and has a half-life of approximately 1-1.5 hours, as measured by fluorescence intensity of cells treated with the protein synthesis inhibitor, cycloheximide.
peTurboGFP-dest1 carries synthetic version of the TurboGFP-dest1 gene which codon usage is optimized for high expression in mammalian cells (humanized) [Haas et al., 1996]. To increase mRNA translation efficiency, Kozak consensus translation initiation site is generated upstream of the TurboGFP-dest1 coding sequence [Kozak, 1987]. Fragments of exons 2 and 3 and intron 2 of human beta globin gene are added in the 3 UTR of TurboGFP-dest1 coding sequence in order to increase the protein expression level.
peTurboGFP-dest1 vector can be used to express TurboGFP-dest1 in eukaryotic (mammalian) cells. For example it can be used as a positive control with a peTurboGFP-PRL-dest1 promoterless vector (Cat.# FP523). The vector can be also used to generate destabilized TurboGFP-tagged fusion proteins. Multiple cloning site (MCS) is located upstream of TurboGFP-dest1 coding sequence.
The vector backbone contains immediate early promoter of cytomegalovirus (PCMV IE) for protein expression, SV40 origin for replication in mammalian cells expressing SV40 T-antigen, pUC origin of replication for propagation in
SV40 early promoter (PSV40) provides neomycin resistance gene (Neor) expression to select stably transfected eukaryotic cells using G418. Bacterial promoter (P) provides kanamycin resistance gene expression (Kanr) in
Generation of TurboGFP-dest1-tagged fusions
A localization signal or a gene of interest can be cloned into MCS of the vector. It will be expressed as a fusion to the TurboGFP-dest1 N-terminus when inserted in the same reading frame as TurboGFP and no in-frame stop codons are present. TurboGFP-dest1-tagged fusions retain fluorescent properties of the native protein allowing fusion localization in vivo. Unmodified vector will express TurboGFP-dest1 when transfected into eukaryotic (mammalian) cells.
Note: The plasmid DNA was isolated from dam+-methylated
Expression in mammalian cells
peTurboGFP-dest1 vector can be transfected into mammalian cells by any known transfection method. CMV promoter provides strong, constitutive expression of TurboGFP-dest1 or its fusions in eukaryotic cells. If required, stable transformants can be selected using G418 [Gorman, 1985].
Propagation in
Suitable host strains for propagation in
Location of features
PCMV IE: 1-589
Enhancer region: 59-465
TATA box: 554-560
Transcription start point: 583
MCS: 591-671
TurboGFP-dest1
Kozak consensus translation initiation site: 672-682
Start codon (ATG): 679-681
Last amino acid in TurboGFP: 1372-1374
Amino acid residues of mouse ornithine decarboxylase (MODC) PEST sequence: 1390-1509
Stop codon: 1510-1512
Fragment of human beta globin (HBB) gene
Last 35 bp of HBB exon 2: 1521-1555
HBB intron 2: 1556-2406
First 233 bp of HBB exon 3: 2407-2639
SV40 early mRNA polyadenylation signal
Polyadenylation signals: 2781-2786 & 2810-2815
mRNA 3' ends: 2819 & 2831
f1 single-strand DNA origin: 2878-3333
Bacterial promoter for expression of Kanr gene
-35 region: 3395-3400
-10 region: 3418-3423
Transcription start point: 3430
SV40 origin of replication: 3674-3809
SV40 early promoter
Enhancer (72-bp tandem repeats): 3507-3578 & 3579-3650
21-bp repeats: 3654-3674, 3675-3695 & 3697-3717
Early promoter element: 3730-3736
Major transcription start points: 3726, 3764, 3770 & 3775
Kanamycin/neomycin resistance gene
Neomycin phosphotransferase coding sequences:
Start codon (ATG): 3858-3860
Stop codon: 4650-4652
G->A mutation to remove Pst I site: 4040
C->A (Arg to Ser) mutation to remove BssH II site: 4386
Herpes simplex virus (HSV) thymidine kinase (TK) polyadenylation signal
Polyadenylation signals: 4888-4893 & 4901-4906
pUC plasmid replication origin: 5237-5880
References:
- Gorman C. High efficiency gene transfer into mammalian cells. In DNA cloning: A Practical Approach, Vol. II. Ed. D. M. Glover. (IRL Press, Oxford, U.K.). 1985; 143-90.
- Haas J, Park EC, Seed B. Codon usage limitation in the expression of HIV-1 envelope glycoprotein. Curr Biol. 1996; 6 (3):315-24. / pmid: 8805248
- Kozak M. An analysis of 5'-noncoding sequences from 699 vertebrate messenger RNAs. Nucleic Acids Res. 1987; 15 (20):8125-48. / pmid: 3313277
- Li X, Zhao X, Fang Y, Jiang X, Duong T, Fan C, Huang CC, Kain SR. Generation of destabilized green fluorescent protein as a transcription reporter. J Biol Chem. 1998; 273 (52):34970-5. / pmid: 9857028
Notice to Purchaser:
TurboGFP-related materials (also referred to as "Products") are intended for research use only. The Products are covered by U.S. Pat. 7,678,893; European Pat. 1576157; and other Evrogen Patents and/or Patent applications pending. By use of these Products, you accept the terms and conditions of the applicable Limited Use Label License.
|
Copyright 2002-2023 Evrogen. All rights reserved. Evrogen JSC, 16/10 Miklukho-Maklaya str., Moscow, Russia, Tel +7(495)988-4084, Fax +7(495)988-4085, e-mail:evrogen@evrogen.com |


