
|
Yellow fluorescent protein TagYFP
- Bright yellow fluorescence
- Monomeric protein with successful performance in fusions
- Fast maturation, high pH-stability and photostability
- Proven suitability to generate stably transfected cell lines
- Recommended for protein labeling
 |
TagYFP is a monomeric yellow fluorescent protein developed on the basis of GFP-like protein from jellyfish Aequorea macrodactyla [Xia et al., 2002]. TagYFP possesses single excitation maximum at 508 nm, and emission maximum at 524 nm. TagYFP is more pH stable than EYFP.
TagYFP is mainly intended for protein labeling in protein localization and interaction studies. It can also be used for cell and organelle labeling and for tracking the promoter activity, although TurboYFP and Phi-Yellow proteins are preferable for such applications because they mature faster and give brighter fluorescent signal.
|
Main properties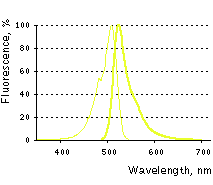
TagYFP normalized excitation (thin line) and emission (thick line) spectra.
Spectra viewer tool
Download TagYFP spectra (xls)
| | CHARACTERISTIC | |
|---|
* Brightness is a product of extinction coefficient and quantum yield, divided by 1000.
| | Molecular weight, kDa | 27.0 | | Polypeptide length, aa | 239 | | Fluorescence color | yellow | | Excitation maximum, nm | 508 | | Emission maximum, nm | 524 | | Quantum yield | 0.62 | | Extinction coefficient, M-1cm-1 | 50 000 | | Brightness* | 31 | | Brightness, % of EGFP | 94 | | pKa | 5.5 | | Structure | monomer | | Aggregation | no | | Maturation rate at 37°C | fast | | Photostability | high | | Cell toxicity | not observed | | Main advantages | bright yellow monomeric fluorescent protein |
|
|---|
Recommended filter sets and antibodies
The protein can be recognized using Anti-Tag(CGY)FP antibody (Cat.# AB121) or Anti-GFP antibody available from Evrogen.
Recommended Omega Optical filter sets for TagYFP are XF104-3 and XF105-2. It can also be detected using Chroma Technology Corp. filter set 41028 Yellow GFP BP (10C/Topaz) or the similar.
Performance and use
TagYFP can be easily expressed and detected in a wide range of organisms. Mammalian cells transiently transfected with TagYFP expression vectors produce bright fluorescence in 10-12 hrs after transfection. No cytotoxic effects or visible protein aggregation are observed.
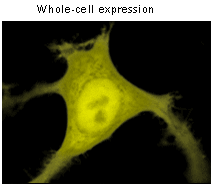 | 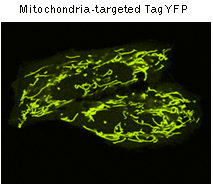 | TagYFP use for labeling of cells and cellular orgenelles.
Confocal microscopy of transiently transfected mammalian cells expressing TagYFP.
|
|---|
TagYFP performance in fusions has been demonstrated in human cytoplasmic β-actin and α-tubulin models. An expected pattern of fluorescence has been obtained in each case.
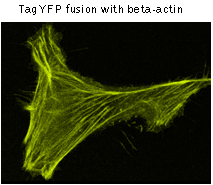 | 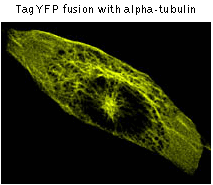 | TagYFP use for protein labeling.
Confocal microscopy of transiently transfected mammalian cells expressing TagYFP fusions.
|
|---|
TagYFP suitability to generate stably transfected cells has been proven by Marinpharm company.
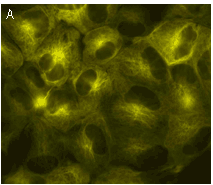 | 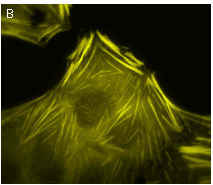 | Stably transfected cell lines expressing TagYFP fusions.
(A) MDCK cells expressing TagYFP-tubulin fusion; (B) U-205 cells expressing TagYFP-actin fusion. Images were kindly provided by Dr. Christian Petzelt (Marinpharm).
|
|---|
TagYFP can be used in multicolor labeling applications with blue, cyan, red, and far-red fluorescent dyes.
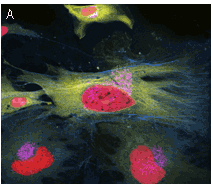 | 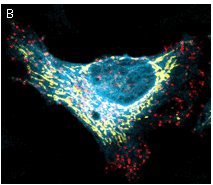 | Multicolor labeling using TagFPs.
(A) TagCFP-tagged β-actin (cyan), TagYFP-tagged α-tubulin (yellow), TagFP635-H2B fusion (red), and Golgi-targeted TagRFP (violet); (B) TagCFP-tagged α-tubulin (cyan), TagFP635-clathrin fusion (red), mitochondria-targeted TagYFP (yellow). Images were kindly provided by Michael W. Davidson (Florida State University).
|
|---|
Available variants and fusions
| Variant | Description | Related vector | Cat.# | Click for image |
|---|
 |
|
Humanized TagYFP
|
TagYFP codon usage is optimized for high expression in mammalian cells [Haas et al., 1996], but it can be successfully expressed in many other heterological systems.
|
pTagYFP-C
|
FP131
|
|
pTagYFP-N
|
FP132
|
|
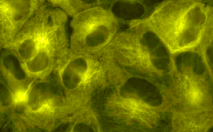
TagYFP-tubulin fusion
|
Human α-tubulin is fused to the TagYFP C-terminus. When expressed in mammalian cells, this fusion provides yellow fluorescent labeling of α-tubulin in living cells.
|
pTagYFP-tubulin
|
FP135
|
|
|
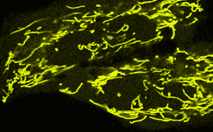
TagYFP-mito fusion
|
A mitochondrial targeting sequence (MTS) is fused to the TagYFP N-terminus. MTS was derived from the subunit VIII of human cytochrome C oxidase [Rizzuto et al., 1989; Rizzuto et al., 1995]. When expressed in mammalian cells, this variant provides yellow fluorescent labeling of mitochondria.
|
pTagYFP-mito
|
FP137
|
|
References:
-
Haas J, Park EC, Seed B.
Codon usage limitation in the expression of HIV-1 envelope glycoprotein.
Curr Biol. 1996; 6 (3):315-24. / pmid: 8805248
-
Rizzuto R, Brini M, Pizzo P, Murgia M, Pozzan T.
Chimeric green fluorescent protein as a tool for visualizing subcellular organelles in living cells.
Curr Biol. 1995; 5 (6):635-42. / pmid: 7552174
-
Rizzuto R, Nakase H, Darras B, Francke U, Fabrizi GM, Mengel T, Walsh F, Kadenbach B, DiMauro S, Schon EA.
A gene specifying subunit VIII of human cytochrome c oxidase is localized to chromosome 11 and is expressed in both muscle and non-muscle tissues.
J Biol Chem. 1989; 264 (18):10595-600. / pmid: 2543673
-
Xia NS, Luo WX, Zhang J, Xie XY, Yang HJ, Li SW, Chen M, Ng MH.
Bioluminescence of Aequorea macrodactyla, a common jellyfish species in the East China Sea.
Mar Biotechnol (NY). 2002; 4 (2):155-62. / pmid: 14961275
|
















