Effects of various conditions on DSN activity.
Activity of DNAse on ds DNA substrate was measured using modified Kunitz assay (Kunitz, 1950)
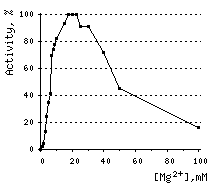
DSN acquires its enzymatic activity in the presence of divalent cations (Mn2+, Co2+, or Mg2+). Mg2+ ion concentration for most applications should be at least 5 mM. DSN is inhibited by EDTA.
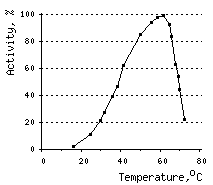
The temperature optimum for DSN activity is 60°C. However, already at 80°C DSN retains only 10% of activity. This sharp decrease in activity may be attributable, at least in part, to ds DNA substrate denaturation.
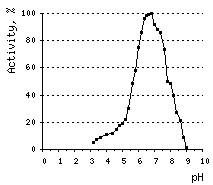
The optimal pH for DSN activity is 6.6. At pH values between 3 and 5, DSN displays only 10% of its maximal activity. The nuclease is stable at a wide range of pH (from 4 to 12) and temperatures below 65°C. About 60% of DSN activity remains after 30-min incubation at 70°C, and 40% – after incubation at 80°C.
 |  |  | |
|---|---|---|---|
Effects of various conditions on DSN activity.Activity of DNAse on ds DNA substrate was measured using modified Kunitz assay (Kunitz, 1950) | |||
Incubation of DSN with aggressive chemicals like 1% SDS, 10 mM β-mercaptoethanol, and 0.3% hydrogen peroxide at 37°C results in only a moderate drop in activity, after 30 min incubation about 90% of activity is preserved. However, a sharp decrease in activity was observed upon chemical treatment at 60°C. SDS completely inhibits DSN activity, while β-mercaptoethanol and hydrogen peroxide induce approximately 70% and 80% loss in activity, respectively.
DSN is highly sensitive to ionic force (e.g., a 10 times decrease in catalytic activity is observed in the presence of 0.2 M NaCl). The addition of chaotropic agents or polyamines to the reaction mixture also results in suppression of enzyme activity.
DSN is tolerant to proteinase K treatment (incubation at 37°C for 30 min).